Robustness of aerosol delivery of amikacin liposome inhalation suspension using the eFlow® Technology - ScienceDirect

Robustness of aerosol delivery of amikacin liposome inhalation suspension using the eFlow® Technology - ScienceDirect
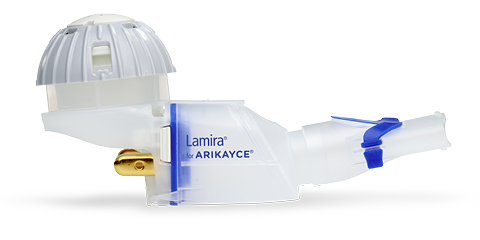
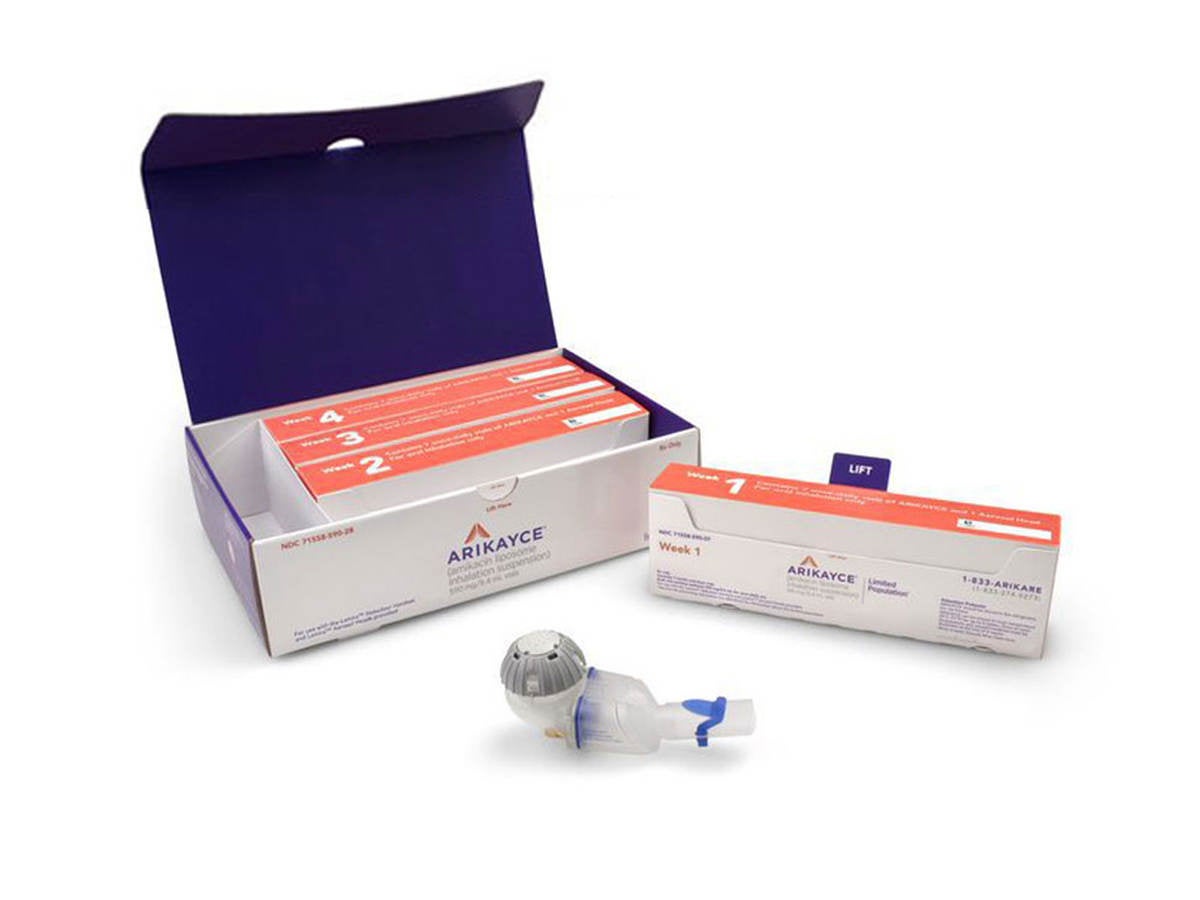
ARIKAYCE® (amikacin liposome inhalation suspension) is the first and only FDA-approved therapy indicated for the treatment of
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ARIKAYCE safely and effe